How to mend a broken heart; heart valve anatomy, disease and treatment
Sep 13, 2022
The Size of the Problem
Cardiovascular disease is the leading cause of death worldwide. The World Health Organisation estimated that 17.9million died from cardiovascular diseases in 2019, which accounted for 32% of all global deaths.
85% of these deaths were due to heart attack or stroke in which the arteries supplying the heart and brain become blocked due to a build-up of fatty deposits over time. Strokes may also occur due to a bleed from the arteries in the brain. Many of these deaths are preventable with lifestyle changes.
Another major cause of cardiovascular death is due to diseases affecting the heart valves.
In this article, you will learn about the heart valve names as well as,
- anatomy of the heart valves and chambers
- heart valves problems
- symptoms of heart valve disease
- management of valvular heart disease
- heart valve replacements
Anatomy of the Heart Chambers and Valves
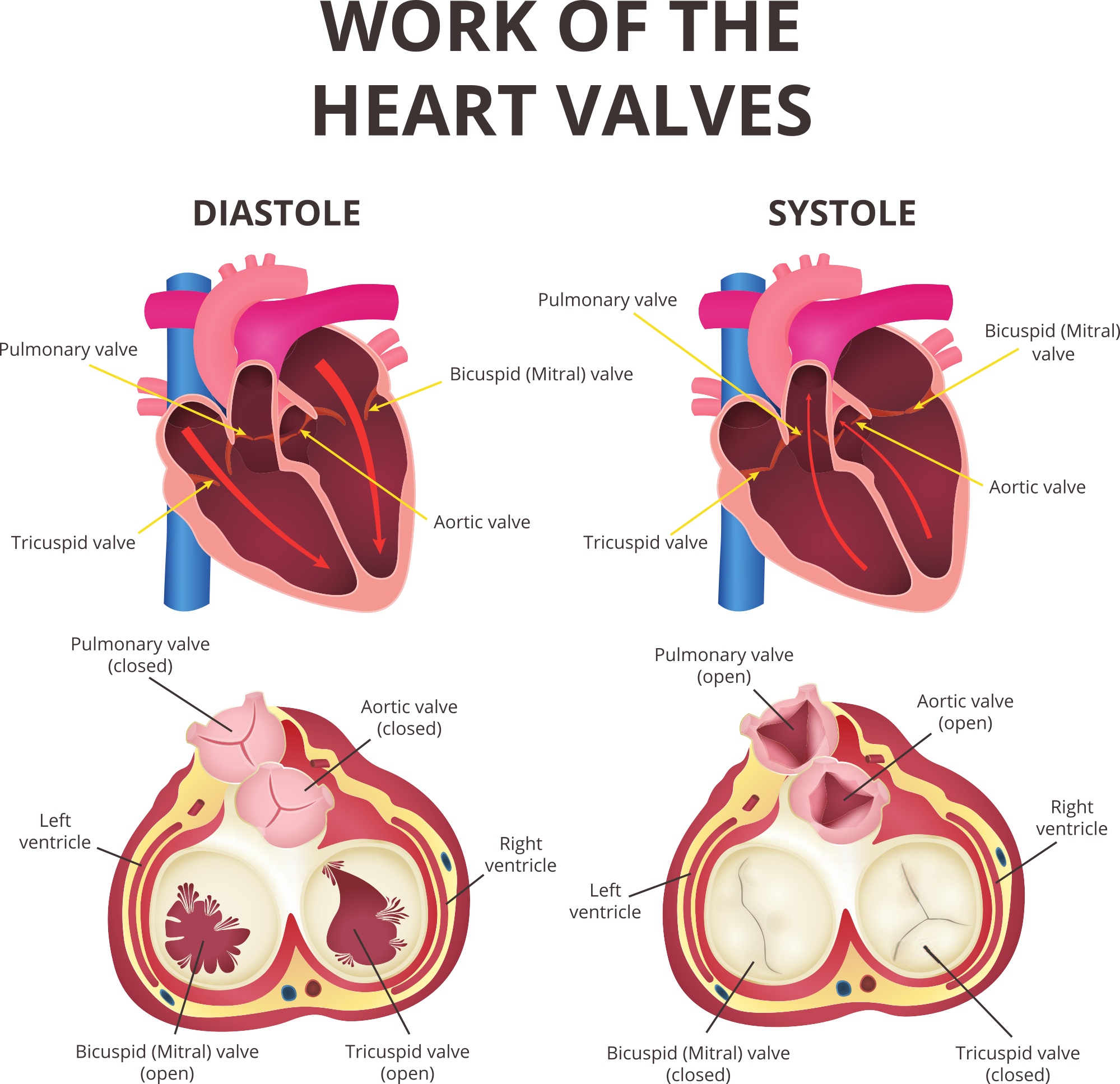
The heart is essentially a biological pump that pushes blood around the body. The heart pumps blood by contraction of powerful cardiac muscles that form the walls of the chambers of the heart. The contraction phase is known as systole, and the relaxation phase is diastole.
Functionally, the heart can be considered in two parts: the right heart and the left heart.
The right heart has two chambers: the right atrium and the right ventricle. The left heart consists of the left atrium and the left ventricle.
The right atrium receives deoxygenated blood from the body. From here, the blood passes into the right ventricle. The right ventricle pumps the deoxygenated blood from the right atrium into the pulmonary circulation and the lungs. The left atrium receives oxygenated blood from the lungs via pulmonary circulation, which then passes into the left ventricle. The left ventricle pumps the oxygenated blood around the whole body.
The right and left atria are separated by a muscular wall known as the interatrial septum. The right and left ventricles are separated by another muscular wall known as the interventricular septum.
Right Atrium
The right atrium receives deoxygenated blood from around the body, which is pumped into the right ventricle through the atrioventricular orifice.
The inside of the right atrium is divided into two continuous spaces by a ridge of tissue known as the crista terminalis, which runs downwards from the superior vena cava to the inferior vena cava. The sulcus terminalis cordis is a vertical groove on the outer wall of the right atrium that corresponds to the inner crista terminalis. The sinus of the venae cava is the space that lies posterior to the crista terminalis. It has smooth thin walls and receives the two venae cavae. The area anterior to the crista terminalis has thicker walls that form ridges by the pectinate muscles on the inner surface.
The right atrium receives blood from
- superior vena cava
- inferior vena cava.
- coronary sinus
The superior vena cava enters the superior part of the right atrium and receives blood from the head, neck, upper limbs and upper trunk. The inferior vena cava enters the floor of the right atrium and receives blood from the whole body below the diaphragm. The inferior vena cava passes through an opening in the diaphragm known as the vena cava hiatus at vertebral level T8.
The heart has its own blood supply via the coronary arteries and cardiac veins. The cardiac veins drain into the coronary sinus, which empties directly into the right atrium.
The right interatrial septum has a depression on its surface, just superior to the opening of the inferior vena cava. This is the fossa ovalis, which is the area of the foramen ovale in the fetus. The foramen ovale is an opening from the right atrium directly into the left atrium. In the fetus, blood bypasses the lungs because the mother’s blood supply provides oxygen for the fetus. At birth, the foramen ovale closes, and blood is pumped to the lungs for oxygenation.
Tricuspid Valve
There is a valve between the right atrium and the right ventricle. This valve has three cusps or leaflets and is called the tricuspid valve. The base of each of the leaflets is secured to the atrioventricular orifice by a fibrous ring. The inner surface of the right ventricle has three distinct ridges of muscle known as the papillary muscles. These are the anterior, posterior and septal papillary muscles that send out thin strands called chordae tendinae that attach to the edges of the tricuspid valve. The leaflets of the tricuspid valve open into the right ventricle during diastole to allow blood to flow from the right atrium into the right ventricle. During ventricular systole, the papillary muscles contract and prevent the leaflets of the tricuspid valve from being blown back into the right atrium and help to keep the valve shut. This prevents blood from flowing back into the right atrium.
Right Ventricle
The right ventricle forms most of the anterior and diaphragmatic surfaces of the heart. The inner surface of the right ventricle has numerous ridges called the trabeculae carneae that attach at both ends to the inner ventricular surface. The papillary muscles are specialised trabeculae carneae that link from the ventricular surface to the leaflets of the tricuspid valve. The conus arteriosus is the outflow part of the right ventricle, which leads to the pulmonary trunk. The walls in this region of the right ventricle are smooth.
Pulmonary Valve
The pulmonary valve lies between the right ventricle and the pulmonary trunk. The pulmonary valve has three semilunar cusps or leaflets. The free edge of each cusp has a thickened portion in the middle, known as the nodule of the semilunar cusp, and thinner lateral parts, known as the lunula of the semilunar cusp. The pulmonary valves open during the right ventricular systole and close during diastole to prevent blood from flowing back into the right ventricle as it fills with blood from the right atrium. The deoxygenated blood then flows through the lungs to remove carbon dioxide and receive oxygen.
Left Atrium
The left atrium forms the posterior surface of the heart. The left atrium has two parts. The posterior part has thin walls and a smooth lining and receives the pulmonary veins. The left atrial appendage or auricle projects from the lateral aspect of the atrium and onto the anterior aspect of the heart. The left auricle has thicker walls that contain the musculi pectinati.
The left atrium receives oxygenated blood from the lungs via pulmonary veins that include;
- left superior pulmonary vein
- right superior pulmonary vein
- left inferior pulmonary vein
- right inferior pulmonary vein
Mitral Valve
The mitral valve separates the left atrium and ventricle. It has two cusps or leaflets and was named mitral as it resembles a bishop’s hat, known as a mitre. The base of the anterior and posterior cusps are secured to the atrioventricular orifice by a fibrous ring. The cusps are further secured by chordae tendineae that extends from the anterior and posterior papillary muscles of the inner surface of the left ventricle.
Left Ventricle
The left ventricle lies anterior to the left atrium and forms part of the anterior surface of the heart as well as the diaphragmatic and left pulmonary surfaces. The left ventricle is conical and forms the apex of the heart. The left ventricle is much thicker than the right ventricle. It is lined with finer trabeculae carneae than the right ventricle. The anterior and posterior papillary muscles of the left ventricle are thicker than the right ventricle.
Aortic Valve
The aortic valve has three semilunar cusps and separates the left ventricle from the aorta. The aortic valve has three semilunar cusps with pocket-like sinuses in the walls. The right and left sinuses contain openings that communicate with the right and left coronary arteries that supply the heart with blood. The posterior sinus does not connect with anything and is called the non-coronary sinus.
Diseases Affecting the Heart Valves
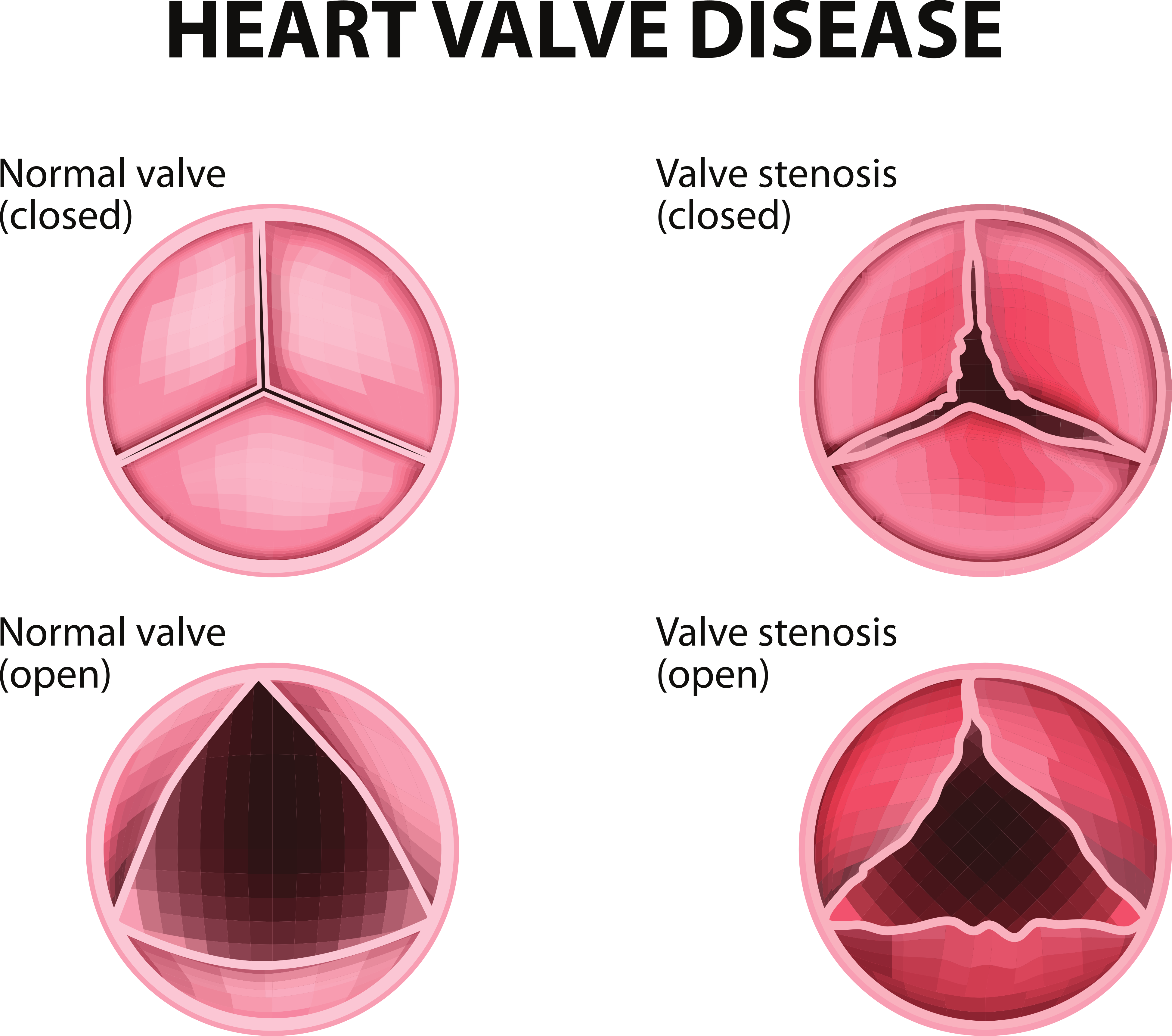
The function of the heart valves may become insufficient either due to thickening and calcification of the leaflets (known as stenosis) or due to increased stretchiness of the tissue (known as prolapse).
In stenosis, the heart valve leaflets may not open fully as they are stiffer. This means that the size of the opening between the heart chambers is reduced, and less blood can flow through it. Over time this causes changes in the blood pressure within the heart. Increased pressure builds up behind the blockage and decreased pressure after the blockage. Stenosed heart valves may not shut properly, which means that blood can flow backwards during systole. This is known as regurgitation.
In a prolapsed valve, the leaflet of the valve is enlarged and flops back into the heart chamber after the valve is shut. This may also cause the backward flow of blood into the heart chamber. Diseases of the aortic valve account for 61% of all valvular heart disease deaths, while diseases of the mitral valve account for 15%
The two most common causes of valve diseases are rheumatic valve disease and degenerative valve disease.
Degenerative Heart Valve Disease
Degenerative valve disease predominantly affects the aortic valve. Calcific deposits form around the valve leaflets making it stiffer. The incidence of aortic valve stenotic disease (AVSD) increases with age, and its prevalence is growing rapidly in high-income countries as people live longer due to better healthcare. Patients with AVSD may present with chest pain (angina), breathlessness (dyspnoea) on exertion, syncope, and heart failure.
Rheumatic Heart Valve disease
Rheumatic valve disease (RVD) is predominant in low-income and middle-income countries. It is associated with overcrowding, poor hygiene, and lack of access to antibiotics. It is far more common in the Indian subcontinent, sub-Saharan Africa, the Middle East, and parts of South America. RVD affects 40.5 million people worldwide, with an annual incidence rate of 2.8 million. Rheumatic valve disease affects younger people, with an estimated 306,000 deaths worldwide, with a median age of 28.7 years. RVD is a complication that occurs after an infection of pharyngitis by group A Streptococcus (Streptococcus pyogenes). Acute rheumatic fever occurs around three weeks after pharyngitis and can affect the joints, skin, brain and heart. It causes fibrosis of the heart valves. Death from RHD may be due to atrial fibrillation (which increases the risk of stroke), pulmonary oedema and heart failure.
Treatment of Valvular Heart Disease
The lining of the heart valves undergoes high shear stresses as the blood flows past them at high pressures. The valves are highly adapted and organised to withstand these pressures.
The mechanical properties of heart valves are maintained by valvular interstitial cells and valvular endothelial cells, which produce a complex network of proteins, including
- collagen
- elastin
- fibronectin
- glycosaminoglycans (GAGs)
- glycoproteins
- proteoglycans (PGs)
These proteins are organised into three distinct layers in the valve; fibrosa, ventricularis and spongiosa
- fibrosa – thickest layer, load bearing predominantly type I collagen
- ventricularis – composed of dens collagen and radially arranged elastin fibres that reduce radial strain during valve opening
- spongiosa – provides lubrication during leaflet bending and contains collagen fibres and hydrated GAGs and PGs
Once the valve is significantly affected by disease, the damage is irreversible, and the only option for treatment is valve replacement surgery.

Heart Valve Replacements
There are two main groups of heart valve replacements: prosthetic heart valves (PHVs) and bio-prosthetic heart valves (BPHVs), also known as tissue valves.
Prosthetic Heart Valves
A PHV is made from a synthetic or manufactured substance with mechanical moving parts. These valves should last a lifetime, but patients must take anticoagulant medication to prevent clots, which increases the risk of bleeding. The design of the valve can be highly regulated to produce accurate pore size, porosity and 3D structure compared to natural tissue valves. However, they may still cause a reaction in the patient and require removal if they get infected. There are different designs of mechanical valves, including ball-in-cage, tilting discs and bi-leaflet valves. PHVs are made from various materials with unique advantages and limitations. These include
- elastomers; polyurethanes, and silicones
- titanium
- pyrolytic carbon
- metal alloys; nickel-titanium, stainless steel
Bio-prosthetic Heart Valves
There are three types of BHPVs
- autografts, harvested from and implanted in the same person
- allografts, human heart valves harvested at post-mortem
- xenografts, animal tissue
The use of xenografts was limited due to immune reactions from the patient rejecting the new valve. These grafts often failed early and had to be revised. Newer preparation techniques, including removing all the cells from the animal tissue before implantation, have been developed to reduce the immune response.
There are three types of BHPV design
- stented – made from porcine or bovine aortic valve tissue sutured onto a polymeric or metal stent
- stentless – made from porcine, bovine or autologous tissue
- percutaneous tissue valves
The Next Frontier - Tissue-Engineered Heart Valves
Tissue engineering is the process of designing 3D functional structures that restore, replace or regenerate damaged body tissues or whole organs. There are three main components of tissue engineering
- 3D scaffold for structure and support
- Cells that can form a biological matrix on the scaffold
- Bioactive molecules such as cytokines and growth factors to stimulate the cells to produce and maintain the biological matrix
The latest advances in tissue-engineered heart valves are either made in vitro or in situ.
The in vitro model includes taking cells from a patient and seeding them onto a 3D scaffold to grow onto the valve in a laboratory before implanting them back into the same patient.
The in-situ approach involves using biological or polymeric valve scaffolds partially seeded with the patient's cells and implanted into the heart to attract new cells. The heart valves will undergo further remodelling after implantation into the patient.
There are two types of scaffolds; either made from synthetic biomaterials or decellularised allogenic or xenogeneic scaffolds. Immune reactions to these valves still occur in vivo, and several new techniques are emerging to decellularise the tissue to mitigate these responses. The chemicals used for these techniques may also cause toxicity to the patient, and the processes need to be further optimised.
Summary
Heart valves perform a vital function to keep the blood circulating smoothly through the different heart chambers. They are highly specialised biological tissues that are constantly maintained by interstitial and endothelial cells to withstand the high shear pressures of the heart. Over a lifetime, the heart can beat 2.5 billion times, and this places great stress on the heart valves. Modern-day lifestyle choices render the heart susceptible to diseases that affect the mechanical function of these valves. Once the valves are damaged, valve replacement surgery is the only option. This type of surgery has been successful but is associated with post-surgical complications, including infections and immune reactions. Tissue engineering techniques are emerging for valve manufacture and preparation to combat these issues and have the potential to offer hope to millions of patients in the future.
Hope you enjoyed learning about heart valves.
I would love to hear from you - drop me a line at info@thefunkyprofessor.com
Stay Funky!
Dr Susan xx
Further reading
World Health Organisation – Fact Sheets
https://www.who.int/en/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
Review of Valvular Heart Disease Epidemiology
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9228968/
Review of Heart Valve Replacements
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7793990/
Review of Imaging of Complications of Cardiac Valve Surgeries
https://doi.org/10.1148/rg.2019180177

